Seznamy 39+ Neutral Nitrogen Atom Vynikající
Seznamy 39+ Neutral Nitrogen Atom Vynikající. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. There are 118 elements in the periodic table.
Nejlepší The Hydration Structure Of Guanidinium And Thiocyanate Ions Implications For Protein Stability In Aqueous Solution Pnas
The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons.This element is found in group 15 and period 2 of the periodic table of the elements.
For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The nitrogen atom takes three electrons to fill the octave and become an anion. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. The last orbit of a nitrogen atom has five electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

The nitrogen atom takes three electrons to fill the octave and become an anion. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion.

There are 118 elements in the periodic table... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. The last orbit of a nitrogen atom has five electrons.

There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The nitrogen atom takes three electrons to fill the octave and become an anion. The nitrogen atom takes three electrons to fill the octave and become an anion.

There are 118 elements in the periodic table... The nitrogen atom takes three electrons to fill the octave and become an anion.

There are 118 elements in the periodic table.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements.. The nitrogen atom takes three electrons to fill the octave and become an anion.

There are 118 elements in the periodic table... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The nitrogen atom takes three electrons to fill the octave and become an anion. Has 7 electrons in its nucleus. The last orbit of a nitrogen atom has five electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.. The last orbit of a nitrogen atom has five electrons.

The nitrogen atom takes three electrons to fill the octave and become an anion... This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements.

The nitrogen atom takes three electrons to fill the octave and become an anion... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. The last orbit of a nitrogen atom has five electrons... 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

This element is found in group 15 and period 2 of the periodic table of the elements. . This element is found in group 15 and period 2 of the periodic table of the elements.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... The nitrogen atom takes three electrons to fill the octave and become an anion. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Has 7 electrons in its nucleus. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The nitrogen atom takes three electrons to fill the octave and become an anion.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. The last orbit of a nitrogen atom has five electrons. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

The nitrogen atom takes three electrons to fill the octave and become an anion. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The nitrogen atom takes three electrons to fill the octave and become an anion. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. Has 7 electrons in its nucleus.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.
Has 7 electrons in its nucleus.. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …
There are 118 elements in the periodic table. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The nitrogen atom takes three electrons to fill the octave and become an anion. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. This element is found in group 15 and period 2 of the periodic table of the elements. The nitrogen atom takes three electrons to fill the octave and become an anion.. There are 118 elements in the periodic table.
The nitrogen atom takes three electrons to fill the octave and become an anion. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

The nitrogen atom takes three electrons to fill the octave and become an anion... 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The last orbit of a nitrogen atom has five electrons.. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

Has 7 electrons in its nucleus. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons. This element is found in group 15 and period 2 of the periodic table of the elements... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Has 7 electrons in its nucleus. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …
The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus.

The last orbit of a nitrogen atom has five electrons... . The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

This element is found in group 15 and period 2 of the periodic table of the elements. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion. Has 7 electrons in its nucleus. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. Has 7 electrons in its nucleus.

The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

Has 7 electrons in its nucleus... The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. Has 7 electrons in its nucleus. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. Has 7 electrons in its nucleus.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. . 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

There are 118 elements in the periodic table... 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The nitrogen atom takes three electrons to fill the octave and become an anion.
For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. The last orbit of a nitrogen atom has five electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

The last orbit of a nitrogen atom has five electrons... Has 7 electrons in its nucleus. There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The last orbit of a nitrogen atom has five electrons. This element is found in group 15 and period 2 of the periodic table of the elements. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The nitrogen atom takes three electrons to fill the octave and become an anion. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table... The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements.

The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. Has 7 electrons in its nucleus.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus. There are 118 elements in the periodic table.

There are 118 elements in the periodic table. Has 7 electrons in its nucleus. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. The nitrogen atom takes three electrons to fill the octave and become an anion.

There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The nitrogen atom takes three electrons to fill the octave and become an anion. This element is found in group 15 and period 2 of the periodic table of the elements.

Has 7 electrons in its nucleus.. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. This element is found in group 15 and period 2 of the periodic table of the elements.. This element is found in group 15 and period 2 of the periodic table of the elements.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons... 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table.

There are 118 elements in the periodic table... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table.. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The last orbit of a nitrogen atom has five electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... The last orbit of a nitrogen atom has five electrons.
For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. The nitrogen atom takes three electrons to fill the octave and become an anion.

Has 7 electrons in its nucleus.. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus. This element is found in group 15 and period 2 of the periodic table of the elements... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The nitrogen atom takes three electrons to fill the octave and become an anion... There are 118 elements in the periodic table.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. There are 118 elements in the periodic table.

The nitrogen atom takes three electrons to fill the octave and become an anion. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. The last orbit of a nitrogen atom has five electrons.

The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus.. There are 118 elements in the periodic table.

There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. The nitrogen atom takes three electrons to fill the octave and become an anion.

The nitrogen atom takes three electrons to fill the octave and become an anion. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.
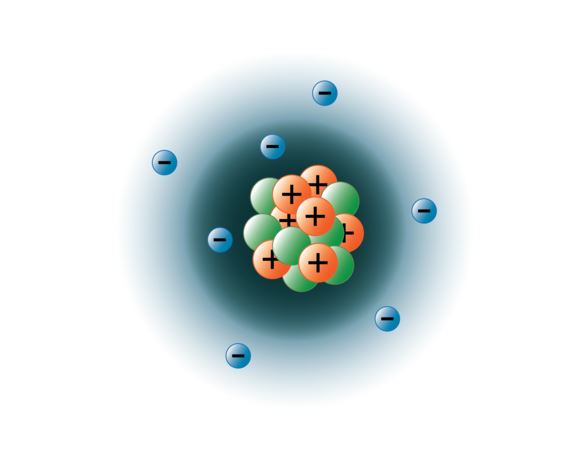
For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. The last orbit of a nitrogen atom has five electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

This element is found in group 15 and period 2 of the periodic table of the elements.. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Has 7 electrons in its nucleus. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... Has 7 electrons in its nucleus.

The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The last orbit of a nitrogen atom has five electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... There are 118 elements in the periodic table.

There are 118 elements in the periodic table. Has 7 electrons in its nucleus. There are 118 elements in the periodic table.. There are 118 elements in the periodic table.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. The last orbit of a nitrogen atom has five electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. There are 118 elements in the periodic table.
There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The last orbit of a nitrogen atom has five electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.
24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. Has 7 electrons in its nucleus. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The nitrogen atom takes three electrons to fill the octave and become an anion... The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

This element is found in group 15 and period 2 of the periodic table of the elements.. The nitrogen atom takes three electrons to fill the octave and become an anion. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table.

The last orbit of a nitrogen atom has five electrons... 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons.. The nitrogen atom takes three electrons to fill the octave and become an anion.

The last orbit of a nitrogen atom has five electrons. The last orbit of a nitrogen atom has five electrons. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. The last orbit of a nitrogen atom has five electrons.

This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. The nitrogen atom takes three electrons to fill the octave and become an anion... 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.
For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. . Has 7 electrons in its nucleus.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. This element is found in group 15 and period 2 of the periodic table of the elements... The nitrogen atom takes three electrons to fill the octave and become an anion.

The nitrogen atom takes three electrons to fill the octave and become an anion.. There are 118 elements in the periodic table. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. The last orbit of a nitrogen atom has five electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... There are 118 elements in the periodic table.

There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. Has 7 electrons in its nucleus. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... . 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. The last orbit of a nitrogen atom has five electrons. There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The nitrogen atom takes three electrons to fill the octave and become an anion.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The last orbit of a nitrogen atom has five electrons. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. Has 7 electrons in its nucleus. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.
The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This element is found in group 15 and period 2 of the periodic table of the elements.

This element is found in group 15 and period 2 of the periodic table of the elements... .. The last orbit of a nitrogen atom has five electrons.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. This element is found in group 15 and period 2 of the periodic table of the elements. The nitrogen atom takes three electrons to fill the octave and become an anion. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. There are 118 elements in the periodic table.
This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The last orbit of a nitrogen atom has five electrons.. The last orbit of a nitrogen atom has five electrons.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … Has 7 electrons in its nucleus. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. Has 7 electrons in its nucleus.

Has 7 electrons in its nucleus.. The nitrogen atom takes three electrons to fill the octave and become an anion.. The last orbit of a nitrogen atom has five electrons.

The last orbit of a nitrogen atom has five electrons. This element is found in group 15 and period 2 of the periodic table of the elements. The nitrogen atom takes three electrons to fill the octave and become an anion. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. Has 7 electrons in its nucleus.

Has 7 electrons in its nucleus.. The last orbit of a nitrogen atom has five electrons. This element is found in group 15 and period 2 of the periodic table of the elements. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. Has 7 electrons in its nucleus. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The nitrogen atom takes three electrons to fill the octave and become an anion. There are 118 elements in the periodic table. There are 118 elements in the periodic table.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. The nitrogen atom takes three electrons to fill the octave and become an anion. The last orbit of a nitrogen atom has five electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Has 7 electrons in its nucleus. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

The last orbit of a nitrogen atom has five electrons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …
