Seznamy 52 Atom Economy Equation Gcse
Seznamy 52 Atom Economy Equation Gcse. The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. Most reactions produce more than one product and very often some of them are not useful.
Prezentováno Aqa Gcse Chemistry Separate Science Unit 3 Quantitative Chemistry Higher
It illustrates what percentage of the mass of. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Atom economy studies the amount of reactants that get turned into useful products. If the theoretical yield is 3.0g, but only 1.4g is produced.%𝑌𝑖 = 𝑥 100 (2 marks) q4:
(2 marks) atom economy q5: It illustrates what percentage of the mass of. Most reactions produce more than one product and very often some of them are not useful. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Of a reaction is a measure of how many reactant atoms form a desired product. Calcium oxide is reacted with water to form calcium hydroxide.

What is the percentage yield?. Why is it important to look at atom economy? Most reactions produce more than one product and very often some of them are not useful. If the theoretical yield is 3.0g, but only 1.4g is produced. (2 marks) atom economy q5: Complete the equation for how atom economy is calculated.. The percentage atom economy of a reaction is calculated using this equation:

%𝑌𝑖 = 𝑥 100 (2 marks) q4:.. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Why is it important to look at atom economy?

Why is it important to look at atom economy? The percentage atom economy of a reaction is calculated using this equation: If the theoretical yield is 3.0g, but only 1.4g is produced. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Why is it important to look at atom economy?. Why is it important to look at atom economy?

It illustrates what percentage of the mass of. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Of a reaction is a measure of how many reactant atoms form a desired product. What is the percentage yield? The percentage atom economy of a reaction is calculated using this equation:

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.. Atom economy studies the amount of reactants that get turned into useful products. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Complete the equation for how atom economy is calculated. Most reactions produce more than one product and very often some of them are not useful... Along with the percentage yield, atom economy is used to analyse the efficiency of reactions.

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Of a reaction is a measure of how many reactant atoms form a desired product. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) (2 marks) atom economy q5: Complete the equation for how atom economy is calculated. It illustrates what percentage of the mass of. Calcium oxide is reacted with water to form calcium hydroxide.. If the theoretical yield is 3.0g, but only 1.4g is produced.

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions... Of a reaction is a measure of how many reactant atoms form a desired product.. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions.

Why is it important to look at atom economy? %𝑌𝑖 = 𝑥 100 (2 marks) q4: Of a reaction is a measure of how many reactant atoms form a desired product. What is the percentage yield? If the theoretical yield is 3.0g, but only 1.4g is produced. It illustrates what percentage of the mass of. Calcium oxide is reacted with water to form calcium hydroxide. Why is it important to look at atom economy? Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. (2 marks) atom economy q5: Atom economy studies the amount of reactants that get turned into useful products... Atom economy studies the amount of reactants that get turned into useful products.

(2 marks) atom economy q5: %𝑌𝑖 = 𝑥 100 (2 marks) q4: What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. Complete the equation for how atom economy is calculated. The percentage atom economy of a reaction is calculated using this equation: Most reactions produce more than one product and very often some of them are not useful. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Atom economy studies the amount of reactants that get turned into useful products.. Of a reaction is a measure of how many reactant atoms form a desired product.

Atom economy studies the amount of reactants that get turned into useful products... Calcium oxide is reacted with water to form calcium hydroxide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Why is it important to look at atom economy? Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. The percentage atom economy of a reaction is calculated using this equation: Atom economy studies the amount of reactants that get turned into useful products. %𝑌𝑖 = 𝑥 100 (2 marks) q4: (2 marks) atom economy q5: If the theoretical yield is 3.0g, but only 1.4g is produced.. Of a reaction is a measure of how many reactant atoms form a desired product.

( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark). (2 marks) atom economy q5: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Calcium oxide is reacted with water to form calcium hydroxide. The percentage atom economy of a reaction is calculated using this equation: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. What is the percentage yield? It illustrates what percentage of the mass of. Calcium oxide is reacted with water to form calcium hydroxide.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. What is the percentage yield? %𝑌𝑖 = 𝑥 100 (2 marks) q4: If the theoretical yield is 3.0g, but only 1.4g is produced. The percentage atom economy of a reaction is calculated using this equation: It illustrates what percentage of the mass of. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark). Of a reaction is a measure of how many reactant atoms form a desired product.

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. If the theoretical yield is 3.0g, but only 1.4g is produced. Atom economy studies the amount of reactants that get turned into useful products. Calcium oxide is reacted with water to form calcium hydroxide. Atom economy studies the amount of reactants that get turned into useful products.

%𝑌𝑖 = 𝑥 100 (2 marks) q4: Atom economy studies the amount of reactants that get turned into useful products. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Why is it important to look at atom economy? Most reactions produce more than one product and very often some of them are not useful.. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) . Most reactions produce more than one product and very often some of them are not useful.

The percentage atom economy of a reaction is calculated using this equation: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. (2 marks) atom economy q5: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. What is the percentage yield? %𝑌𝑖 = 𝑥 100 (2 marks) q4:. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

Most reactions produce more than one product and very often some of them are not useful. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. The percentage atom economy of a reaction is calculated using this equation: %𝑌𝑖 = 𝑥 100 (2 marks) q4: It illustrates what percentage of the mass of. Of a reaction is a measure of how many reactant atoms form a desired product. (2 marks) atom economy q5: What is the percentage yield?

%𝑌𝑖 = 𝑥 100 (2 marks) q4: It illustrates what percentage of the mass of. Most reactions produce more than one product and very often some of them are not useful. Why is it important to look at atom economy? Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. %𝑌𝑖 = 𝑥 100 (2 marks) q4:.. If the theoretical yield is 3.0g, but only 1.4g is produced.

Atom economy studies the amount of reactants that get turned into useful products. Of a reaction is a measure of how many reactant atoms form a desired product. What is the percentage yield? Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Complete the equation for how atom economy is calculated. Why is it important to look at atom economy? ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Atom economy studies the amount of reactants that get turned into useful products. Complete the equation for how atom economy is calculated.

Atom economy studies the amount of reactants that get turned into useful products.. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. It illustrates what percentage of the mass of. Most reactions produce more than one product and very often some of them are not useful. Of a reaction is a measure of how many reactant atoms form a desired product. Calcium oxide is reacted with water to form calcium hydroxide. Why is it important to look at atom economy? Complete the equation for how atom economy is calculated.. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

Of a reaction is a measure of how many reactant atoms form a desired product. . (2 marks) atom economy q5:

Complete the equation for how atom economy is calculated. The percentage atom economy of a reaction is calculated using this equation: Complete the equation for how atom economy is calculated. What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Why is it important to look at atom economy? Calcium oxide is reacted with water to form calcium hydroxide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Most reactions produce more than one product and very often some of them are not useful. %𝑌𝑖 = 𝑥 100 (2 marks) q4: The percentage atom economy of a reaction is calculated using this equation:

Calcium oxide is reacted with water to form calcium hydroxide... Most reactions produce more than one product and very often some of them are not useful. Complete the equation for how atom economy is calculated. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Calcium oxide is reacted with water to form calcium hydroxide. Atom economy studies the amount of reactants that get turned into useful products. What is the percentage yield? Of a reaction is a measure of how many reactant atoms form a desired product. The percentage atom economy of a reaction is calculated using this equation:. Calcium oxide is reacted with water to form calcium hydroxide.

Of a reaction is a measure of how many reactant atoms form a desired product... Atom economy studies the amount of reactants that get turned into useful products. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Most reactions produce more than one product and very often some of them are not useful. Complete the equation for how atom economy is calculated. The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. If the theoretical yield is 3.0g, but only 1.4g is produced. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Of a reaction is a measure of how many reactant atoms form a desired product. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Most reactions produce more than one product and very often some of them are not useful. What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. (2 marks) atom economy q5: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. %𝑌𝑖 = 𝑥 100 (2 marks) q4:.. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark)

What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. Of a reaction is a measure of how many reactant atoms form a desired product. Calcium oxide is reacted with water to form calcium hydroxide. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) %𝑌𝑖 = 𝑥 100 (2 marks) q4: Complete the equation for how atom economy is calculated. Why is it important to look at atom economy? It illustrates what percentage of the mass of.

Complete the equation for how atom economy is calculated. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Atom economy studies the amount of reactants that get turned into useful products. The percentage atom economy of a reaction is calculated using this equation: Most reactions produce more than one product and very often some of them are not useful.

It illustrates what percentage of the mass of.. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. %𝑌𝑖 = 𝑥 100 (2 marks) q4: What is the percentage yield? Calcium oxide is reacted with water to form calcium hydroxide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. It illustrates what percentage of the mass of. Why is it important to look at atom economy?. Calcium oxide is reacted with water to form calcium hydroxide.

It illustrates what percentage of the mass of. Of a reaction is a measure of how many reactant atoms form a desired product... Of a reaction is a measure of how many reactant atoms form a desired product.

It illustrates what percentage of the mass of. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. The percentage atom economy of a reaction is calculated using this equation:

Of a reaction is a measure of how many reactant atoms form a desired product. Atom economy studies the amount of reactants that get turned into useful products. Complete the equation for how atom economy is calculated. (2 marks) atom economy q5: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. It illustrates what percentage of the mass of. The percentage atom economy of a reaction is calculated using this equation: Of a reaction is a measure of how many reactant atoms form a desired product.. What is the percentage yield?

If the theoretical yield is 3.0g, but only 1.4g is produced... Complete the equation for how atom economy is calculated. The percentage atom economy of a reaction is calculated using this equation: Why is it important to look at atom economy?.. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark)

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Why is it important to look at atom economy? If the theoretical yield is 3.0g, but only 1.4g is produced. It illustrates what percentage of the mass of. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Complete the equation for how atom economy is calculated. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. %𝑌𝑖 = 𝑥 100 (2 marks) q4:.. If the theoretical yield is 3.0g, but only 1.4g is produced.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. %𝑌𝑖 = 𝑥 100 (2 marks) q4:. Most reactions produce more than one product and very often some of them are not useful.

Calcium oxide is reacted with water to form calcium hydroxide... Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Of a reaction is a measure of how many reactant atoms form a desired product. If the theoretical yield is 3.0g, but only 1.4g is produced. What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. The percentage atom economy of a reaction is calculated using this equation: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Calcium oxide is reacted with water to form calcium hydroxide. (2 marks) atom economy q5: Why is it important to look at atom economy?.. What is the percentage yield?

Calcium oxide is reacted with water to form calcium hydroxide. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) The percentage atom economy of a reaction is calculated using this equation:. Most reactions produce more than one product and very often some of them are not useful.
The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. If the theoretical yield is 3.0g, but only 1.4g is produced. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. It illustrates what percentage of the mass of. What is the percentage yield? Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide... (2 marks) atom economy q5:

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions... Of a reaction is a measure of how many reactant atoms form a desired product.

It illustrates what percentage of the mass of. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions... It illustrates what percentage of the mass of.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide... Most reactions produce more than one product and very often some of them are not useful. It illustrates what percentage of the mass of. What is the percentage yield? If the theoretical yield is 3.0g, but only 1.4g is produced. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Complete the equation for how atom economy is calculated. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Atom economy studies the amount of reactants that get turned into useful products. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. (2 marks) atom economy q5:

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. .. It illustrates what percentage of the mass of.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. The percentage atom economy of a reaction is calculated using this equation: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Of a reaction is a measure of how many reactant atoms form a desired product. The percentage atom economy of a reaction is calculated using this equation:
Most reactions produce more than one product and very often some of them are not useful. Most reactions produce more than one product and very often some of them are not useful. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) It illustrates what percentage of the mass of. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. (2 marks) atom economy q5: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Calcium oxide is reacted with water to form calcium hydroxide. Why is it important to look at atom economy? ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark)

It illustrates what percentage of the mass of... ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) %𝑌𝑖 = 𝑥 100 (2 marks) q4: (2 marks) atom economy q5:.. Calcium oxide is reacted with water to form calcium hydroxide.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. What is the percentage yield? %𝑌𝑖 = 𝑥 100 (2 marks) q4: Complete the equation for how atom economy is calculated. Most reactions produce more than one product and very often some of them are not useful. The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. Why is it important to look at atom economy? It illustrates what percentage of the mass of... Complete the equation for how atom economy is calculated.

Atom economy studies the amount of reactants that get turned into useful products... (2 marks) atom economy q5: Of a reaction is a measure of how many reactant atoms form a desired product. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. What is the percentage yield?.. (2 marks) atom economy q5:

%𝑌𝑖 = 𝑥 100 (2 marks) q4:.. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Why is it important to look at atom economy? Of a reaction is a measure of how many reactant atoms form a desired product. Complete the equation for how atom economy is calculated. (2 marks) atom economy q5: Most reactions produce more than one product and very often some of them are not useful. It illustrates what percentage of the mass of. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.. It illustrates what percentage of the mass of.

Complete the equation for how atom economy is calculated. Most reactions produce more than one product and very often some of them are not useful. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Complete the equation for how atom economy is calculated. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. %𝑌𝑖 = 𝑥 100 (2 marks) q4:. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark)
Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. (2 marks) atom economy q5: Why is it important to look at atom economy? %𝑌𝑖 = 𝑥 100 (2 marks) q4: Atom economy studies the amount of reactants that get turned into useful products.

If the theoretical yield is 3.0g, but only 1.4g is produced. The percentage atom economy of a reaction is calculated using this equation: If the theoretical yield is 3.0g, but only 1.4g is produced. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Most reactions produce more than one product and very often some of them are not useful.

If the theoretical yield is 3.0g, but only 1.4g is produced.. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Complete the equation for how atom economy is calculated.. If the theoretical yield is 3.0g, but only 1.4g is produced.

What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. What is the percentage yield? Complete the equation for how atom economy is calculated. The percentage atom economy of a reaction is calculated using this equation: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Calcium oxide is reacted with water to form calcium hydroxide. Why is it important to look at atom economy? Of a reaction is a measure of how many reactant atoms form a desired product. If the theoretical yield is 3.0g, but only 1.4g is produced. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions.. Atom economy studies the amount of reactants that get turned into useful products.

%𝑌𝑖 = 𝑥 100 (2 marks) q4:. What is the percentage yield? Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Calcium oxide is reacted with water to form calcium hydroxide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. Most reactions produce more than one product and very often some of them are not useful. The percentage atom economy of a reaction is calculated using this equation: Why is it important to look at atom economy?.. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

%𝑌𝑖 = 𝑥 100 (2 marks) q4:. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. What is the percentage yield?. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions.

Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. It illustrates what percentage of the mass of. Complete the equation for how atom economy is calculated. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) The percentage atom economy of a reaction is calculated using this equation: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Calcium oxide is reacted with water to form calcium hydroxide. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

It illustrates what percentage of the mass of.. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Along with the percentage yield, atom economy is used to analyse the efficiency of reactions... (2 marks) atom economy q5:

( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Atom economy studies the amount of reactants that get turned into useful products. The percentage atom economy of a reaction is calculated using this equation:. Complete the equation for how atom economy is calculated.

Most reactions produce more than one product and very often some of them are not useful. .. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

Most reactions produce more than one product and very often some of them are not useful... Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. %𝑌𝑖 = 𝑥 100 (2 marks) q4: (2 marks) atom economy q5: The percentage atom economy of a reaction is calculated using this equation: Calcium oxide is reacted with water to form calcium hydroxide. It illustrates what percentage of the mass of. Atom economy studies the amount of reactants that get turned into useful products... Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide.

Of a reaction is a measure of how many reactant atoms form a desired product. Of a reaction is a measure of how many reactant atoms form a desired product. Most reactions produce more than one product and very often some of them are not useful. Why is it important to look at atom economy?.. If the theoretical yield is 3.0g, but only 1.4g is produced.
What is the percentage yield? Atom economy studies the amount of reactants that get turned into useful products. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Calcium oxide is reacted with water to form calcium hydroxide. If the theoretical yield is 3.0g, but only 1.4g is produced... Atom economy studies the amount of reactants that get turned into useful products.

( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Of a reaction is a measure of how many reactant atoms form a desired product. It illustrates what percentage of the mass of. Calcium oxide is reacted with water to form calcium hydroxide. If the theoretical yield is 3.0g, but only 1.4g is produced. Most reactions produce more than one product and very often some of them are not useful. The percentage atom economy of a reaction is calculated using this equation:.. Of a reaction is a measure of how many reactant atoms form a desired product.

Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide... It illustrates what percentage of the mass of. Why is it important to look at atom economy? What is the percentage yield? %𝑌𝑖 = 𝑥 100 (2 marks) q4: ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. (2 marks) atom economy q5: Of a reaction is a measure of how many reactant atoms form a desired product. Calcium oxide is reacted with water to form calcium hydroxide.. %𝑌𝑖 = 𝑥 100 (2 marks) q4:

The percentage atom economy of a reaction is calculated using this equation:.. Of a reaction is a measure of how many reactant atoms form a desired product. Calcium oxide is reacted with water to form calcium hydroxide. Atom economy studies the amount of reactants that get turned into useful products. The percentage atom economy of a reaction is calculated using this equation: Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. If the theoretical yield is 3.0g, but only 1.4g is produced. It illustrates what percentage of the mass of. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark) (2 marks) atom economy q5:.. ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark)

Why is it important to look at atom economy? Complete the equation for how atom economy is calculated. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. Along with the percentage yield, atom economy is used to analyse the efficiency of reactions. Atom economy studies the amount of reactants that get turned into useful products. %𝑌𝑖 = 𝑥 100 (2 marks) q4: Of a reaction is a measure of how many reactant atoms form a desired product... Atom economy studies the amount of reactants that get turned into useful products.
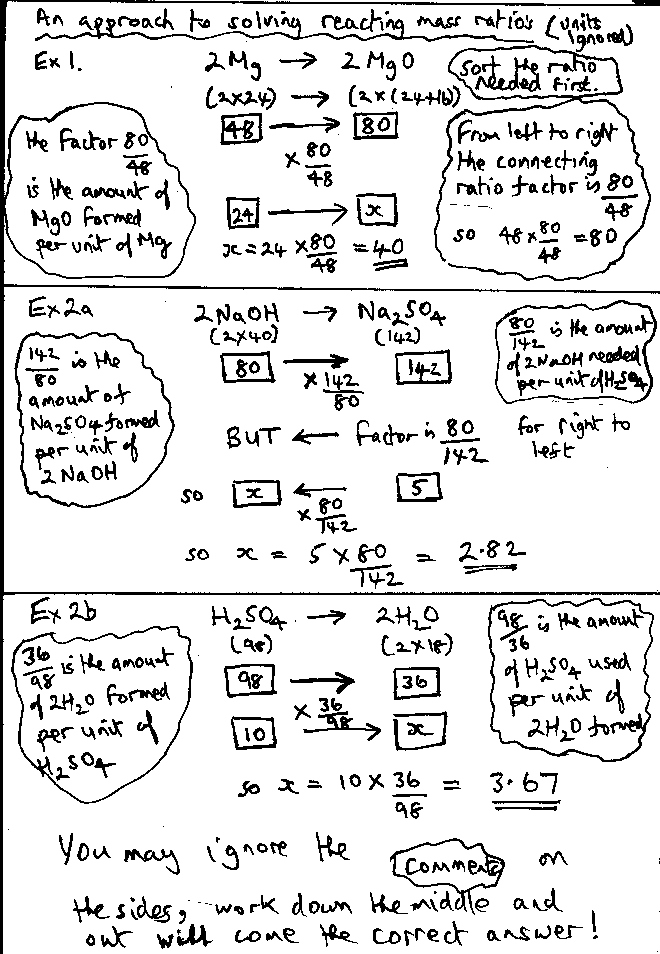
Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide... What is the percentage yield? Complete the equation for how atom economy is calculated. If the theoretical yield is 3.0g, but only 1.4g is produced. Learn about and revise atom economy, percentage yield and gas calculations with this bbc bitesize gcse chemistry (aqa) study guide. It illustrates what percentage of the mass of. (2 marks) atom economy q5: The percentage atom economy of a reaction is calculated using this equation: ( h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o𝑖 n l n k p n k i m q𝑎 p𝑖 k j) i n h𝑎 p𝑖 k n i q h𝑎 𝑎 o o o 𝑎 h h n 𝑎 p𝑎 j p o n k i m q𝑎 p𝑖 k j (1 𝑎 n g) t 100 (1 mark). Of a reaction is a measure of how many reactant atoms form a desired product.
